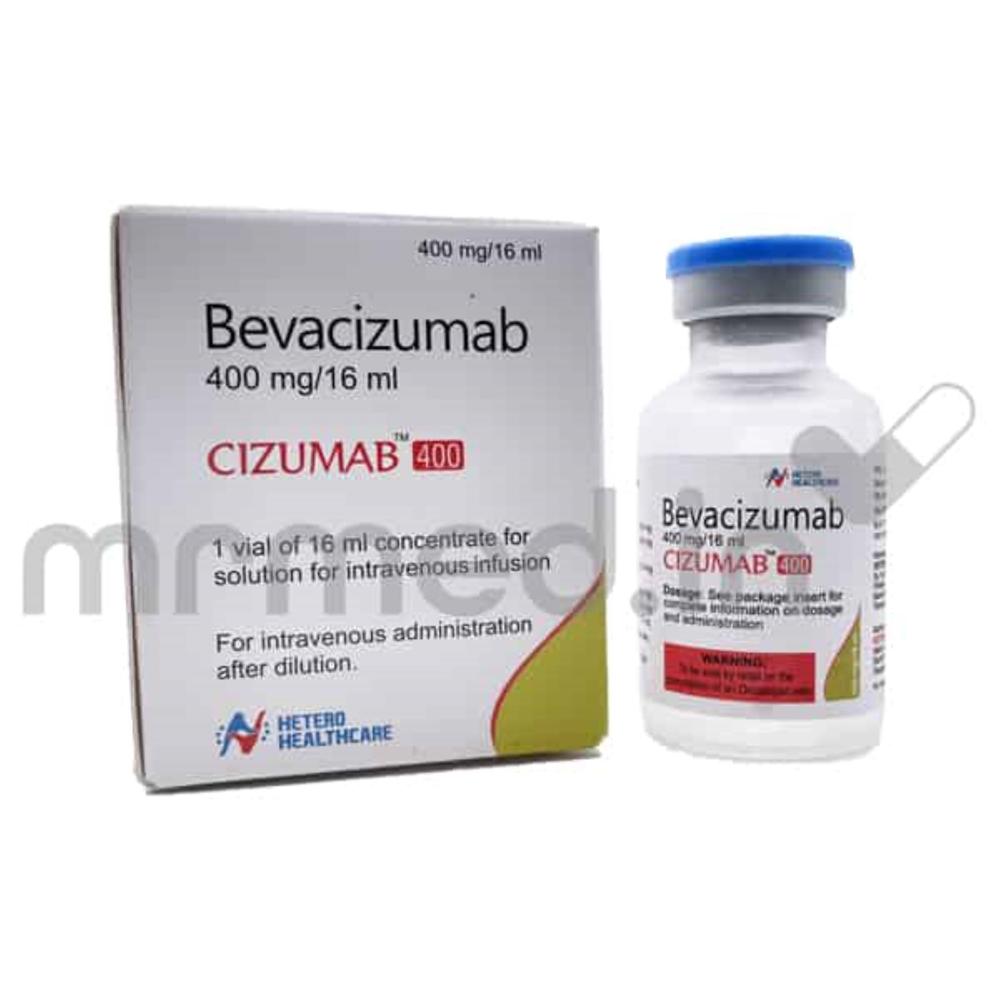
CIZUMAB 400MG INJ
7600.0 INR/Number
Product Details:
- Packaging Type Vial
- Pacakaging (Quantity Per Box) 1 Vial per box
- Life Span 36 months from date of manufacture
- Indication Metastatic Colorectal Cancer, Non-Small Cell Lung Cancer, Metastatic Breast Cancer, Glioblastoma, Renal Cell Carcinoma, Cervical Cancer
- Dosage Form Injection
- Brand Name CIZUMAB 400MG INJ
- Origin of Medicine India
- Click to View more
X
CIZUMAB 400MG INJ Price And Quantity
- 7600.0 INR/Number
- 1 Number
- Prescription only
- L01XC07
- Hypertension, proteinuria, bleeding, impaired wound healing, gastrointestinal perforation
- FDA/Indian DCGI Approval
- Use with caution in patients with history of thromboembolism, recent surgery, hypertension
- Hypersensitivity to bevacizumab or excipients, pregnancy
- Clear to slightly opalescent, colorless to pale brown liquid
- Available on packaging
- Intravenous (IV)
- Sterile single-use vial
- Infusion should be prepared and administered by healthcare professionals
CIZUMAB 400MG INJ Product Specifications
- Metastatic Colorectal Cancer, Non-Small Cell Lung Cancer, Metastatic Breast Cancer, Glioblastoma, Renal Cell Carcinoma, Cervical Cancer
- Biological/Monoclonal Antibody
- As directed by the oncologist, typically based on body weight or body surface area
- Injection
- India
- CIZUMAB 400MG INJ
- Administered as an intravenous infusion under medical supervision
- Bevacizumab 400 mg / 16 ml
- Store in a refrigerator (2C - 8C); Do not freeze
- Anticancer / Targeted Therapy (Anti-VEGF monoclonal antibody)
- Cancer Treatment (e.g., colorectal, lung, glioblastoma, kidney, and cervical cancer)
- Liquid Injection
- Vial
- Bevacizumab
- 1 Vial of 16 ml
- 1 Vial per box
- Adults
- 36 months from date of manufacture
- Prescription only
- L01XC07
- Hypertension, proteinuria, bleeding, impaired wound healing, gastrointestinal perforation
- FDA/Indian DCGI Approval
- Use with caution in patients with history of thromboembolism, recent surgery, hypertension
- Hypersensitivity to bevacizumab or excipients, pregnancy
- Clear to slightly opalescent, colorless to pale brown liquid
- Available on packaging
- Intravenous (IV)
- Sterile single-use vial
- Infusion should be prepared and administered by healthcare professionals
CIZUMAB 400MG INJ Trade Information
- 1000 Number Per Week
- 2 Days
- No
- Asia, Western Europe, Middle East, Africa, Australia, South America, Eastern Europe, Central America, North America
- All India
Product Description
Cizumab is a recombinant humanized monoclonal antibody that functions as an anti-angiogenic agent. It works by:- Targeting VEGF:It selectively binds to a protein in the body called vascular endothelial growth factor (VEGF).
- Inhibiting Blood Vessel Growth:VEGF usually stimulates the formation of new blood vessels (angiogenesis) that supply oxygen and nutrients to tumors, allowing them to grow and spread.
- Starving the Tumor:By blocking the function of VEGF, Cizumab cuts off the tumor's blood supply, which in turn slows its growth, causes it to shrink, or induces cell death.
Key Uses
Cizumab 400 mg injection is primarily used to treat several types of cancer, usually in combination with other cancer therapies:
- Metastatic cancer of the colon and rectum
- Advanced non-small cell lung cancer
- Kidney cancer (renal cell carcinoma)
- Brain tumors (glioblastoma multiforme)
- Ovarian, fallopian tube, or primary peritoneal cancer
- Metastatic cervical cancer
- Liver cancer (hepatocellular carcinoma)
Targeted Cancer Therapy
CIZUMAB 400MG INJ employs bevacizumab, a biological agent that inhibits vascular endothelial growth factor (VEGF), helping to prevent the growth of blood vessels that feed tumors. This targeted mechanism makes it an effective therapy across multiple types of cancers including colorectal, lung, brain, kidney, and cervical cancers, offering renewed hope for patients with advanced-stage malignancies.
Administration and Supervision
This injection is strictly administered as an intravenous infusion by trained healthcare professionals in a hospital or clinical setting. The dosage is tailored based on individual body weight or surface area, and patient monitoring is essential during and after infusion to manage potential side effects and ensure optimal therapeutic outcomes.
Safety and Handling Precautions
CIZUMAB 400MG INJ should not be used in individuals with known hypersensitivity to bevacizumab, during pregnancy, or in those with certain pre-existing medical conditions unless directed by an oncologist. Adherence to recommended storage (refrigeration at 2C - 8C) and handling instructions is crucial to maintain product efficacy and safety.
FAQ's of CIZUMAB 400MG INJ:
Q: How is CIZUMAB 400MG INJ administered?
A: CIZUMAB 400MG INJ is given as an intravenous infusion, usually in a hospital or infusion center. Only qualified healthcare professionals should prepare and administer this medication to ensure accurate dosing and immediate management of side effects.Q: What types of cancer is CIZUMAB 400MG INJ used to treat?
A: This injection is used for multiple cancers, including metastatic colorectal cancer, non-small cell lung cancer, metastatic breast cancer, glioblastoma, renal cell carcinoma, and cervical cancer as recommended by your oncologist.Q: When should CIZUMAB 400MG INJ not be used?
A: CIZUMAB should not be used in patients with known hypersensitivity to bevacizumab or its excipients, during pregnancy, or in those where the physician evaluates significant risk based on medical history, such as recent surgery or uncontrolled hypertension.Q: Where should CIZUMAB 400MG INJ be stored?
A: The vial should be stored in a refrigerator between 2C and 8C and should not be frozen. Always keep the vial in its original packaging to protect it from light until it is ready for use.Q: What are the common side effects of CIZUMAB 400MG INJ?
A: Potential side effects include hypertension, proteinuria (protein in urine), increased risk of bleeding, slowed wound healing, and, rarely, gastrointestinal perforation. Regular monitoring by healthcare professionals helps manage and mitigate these risks.Q: How does CIZUMAB 400MG INJ benefit cancer patients?
A: By inhibiting VEGF, CIZUMAB 400MG INJ prevents the growth of new blood vessels that support tumor growth, effectively slowing or reducing the progression of certain cancers and potentially improving patient survival rates.Q: What process is involved in receiving CIZUMAB 400MG INJ treatment?
A: Treatment involves a prescribed number of intravenous infusions, typically scheduled every two to three weeks. The healthcare team calculates individualized doses, monitors for effectiveness and safety, and adjusts the regimen as necessary.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email


 Contact Us
Contact Us Call Me Free
Call Me Free
