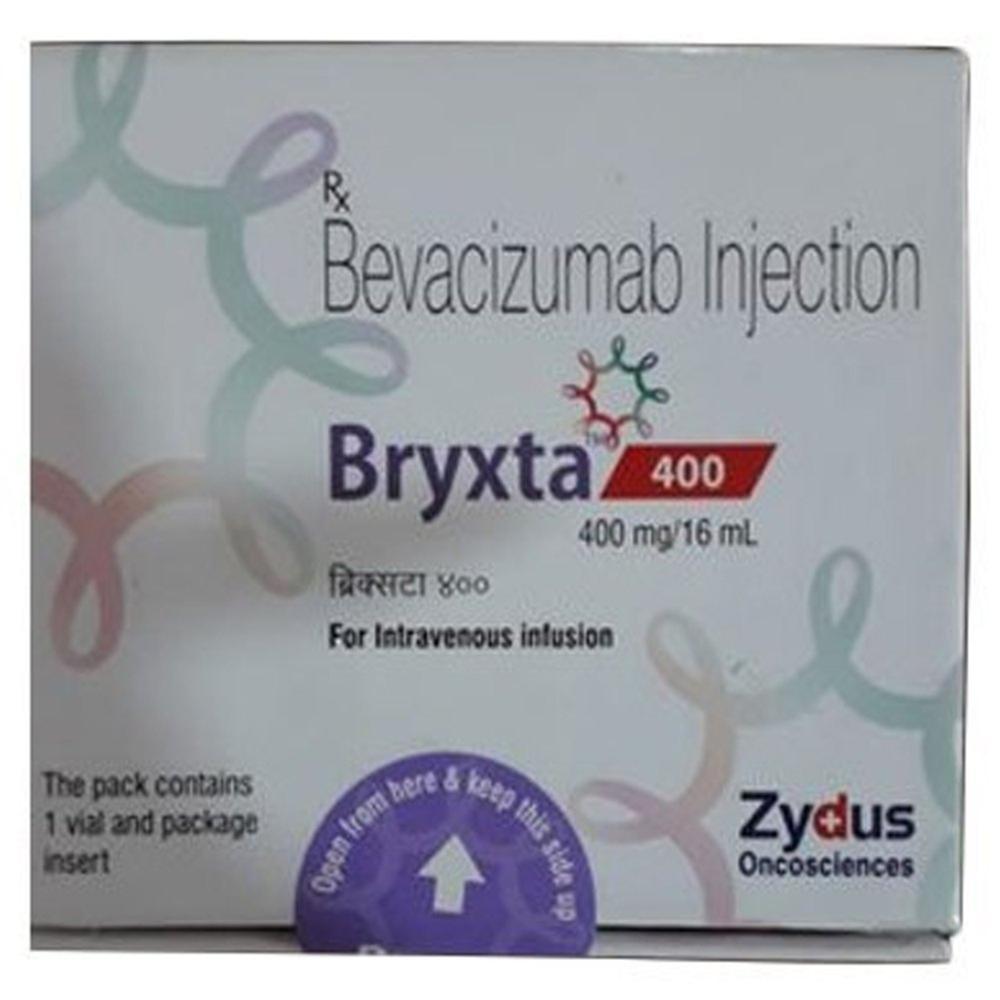
BRYXTA 400MG INJ
7500.0 INR/Number
Product Details:
- Origin of Medicine India
- Life Span 24 months from date of manufacture
- Dosage Form Lyophilized Powder for Solution for Injection
- Indication HER2-positive metastatic breast cancer, HER2-positive metastatic gastric cancer
- Salt Composition Trastuzumab
- Pacakaging (Quantity Per Box) 1 Vial per box
- Brand Name Bryxta 400mg Inj
- Click to View more
X
BRYXTA 400MG INJ Price And Quantity
- 1 Number
- 7500.0 INR/Number
BRYXTA 400MG INJ Product Specifications
- Prescription
- Store in a refrigerator (2C - 8C). Do not freeze.
- Trastuzumab
- HER2-positive metastatic breast cancer, HER2-positive metastatic gastric cancer
- Anti-cancer, Monoclonal Antibody Therapy
- 1 Vial per box
- Injection
- Vial
- Bryxta 400mg Inj
- Patients with HER2-positive breast cancer and gastric cancer
- 1 Vial
- India
- Trastuzumab Lyophilized Powder
- Lyophilized Powder for Solution for Injection
- 24 months from date of manufacture
- As directed by physician
- Adults
- Administered intravenously under medical supervision
- C6470H10012N1724O2013S42
- MNB/19/1017
- Oncology (Antineoplastic)
- 148 kDa
- Zydus Cadila
- Yes (with supplied sterile water for injection)
- Yes
- Intravenous infusion
BRYXTA 400MG INJ Trade Information
- 1000 Number Per Week
- 2 Days
- No
- Middle East, Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Africa
- All India
Product Description
- Active Ingredient: Bevacizumab 400 mg.
- Form: Solution for intravenous (IV) infusion, administered by a healthcare professional in a clinical setting.
- Mechanism of Action: It binds to a protein called vascular endothelial growth factor (VEGF), which is essential for the creation of blood vessels within tumors (angiogenesis). By blocking VEGF, the medicine limits the blood flow to cancer cells, causing the tumor to shrink or stop growing.
- Metastatic colorectal cancer (cancer of the colon or rectum that has spread)
- Advanced non-small cell lung cancer
- Metastatic breast cancer (usually HER2-negative tumors)
- Advanced kidney cancer (renal cell carcinoma)
- Cervical cancer (persistent, recurrent, or metastatic)
- Ovarian, fallopian tube, or primary peritoneal cancer
- Glioblastoma (a type of brain tumor, typically in recurrent cases)
- Hepatocellular carcinoma (liver cancer)
- Administration: This medication must not be self-administered. It is given as an intravenous infusion by a doctor or nurse.
- Surgery: Bryxta can impair wound healing and increase the risk of bleeding. Patients should not have major surgery for at least 28 days before or after receiving the injection, or until the wound has fully healed.
- Pregnancy/Breastfeeding: It is contraindicated during pregnancy and breastfeeding. Women of childbearing potential must use effective contraception during treatment and for at least six months after the final dose.
- Common Side Effects: Include fatigue, constipation, loss of appetite, fever, nosebleeds, headache, dry skin, high blood pressure, and changes in speech or taste.
- Serious Side Effects: Can include severe bleeding (e.g., in the stomach or intestines), blood clots, high blood pressure, protein in the urine, and heart problems. Patients should seek immediate medical attention for symptoms like severe headache, vision changes, confusion, or unusual bleeding.
Uses
Bryxta 400mg is used, often in combination with other chemotherapy drugs, to treat several types of cancer, including:
Important Precautions and Side Effects
Comprehensive Cancer Treatment
BRYXTA 400MG INJ offers advanced monoclonal antibody therapy specifically for patients with HER2-positive metastatic breast and gastric cancers. Its targeted mechanism helps inhibit the growth of cancer cells, bringing hope and improved outcomes in oncology care.
Strict Quality Control and Preservation
Produced by Zydus Cadila under the license MNB/19/1017, BRYXTA is preservative-free to minimize sensitivities. Each vial undergoes rigorous quality checks and should be stored refrigerated (2C - 8C) and never frozen to maintain its potency and shelf life.
Patient-Centric and Physician-Guided Usage
BRYXTA 400MG INJ is reconstituted with sterile water and administered as an intravenous infusion under strict medical supervision. Physicians determine the exact protocol based on the patient's specific case, ensuring both safety and efficacy during oncological treatment.
FAQ's of BRYXTA 400MG INJ:
Q: How is BRYXTA 400MG INJ prepared and administered?
A: BRYXTA 400MG INJ requires reconstitution with the supplied sterile water for injection to create a solution, which is then administered as an intravenous infusion. This process must be performed by trained medical professionals in a hospital or oncology clinic.Q: What are the main indications for using BRYXTA 400MG INJ?
A: The injection is recommended for adults diagnosed with HER2-positive metastatic breast cancer or HER2-positive metastatic gastric cancer. It is used as part of an anti-cancer monoclonal antibody therapy regimen.Q: When should BRYXTA 400MG INJ be taken?
A: BRYXTA 400MG INJ is administered on a schedule determined by your physician, often in cycles as part of a broader chemotherapy plan. Timing and frequency depend on your cancer type, stage, and overall health.Q: Where should BRYXTA 400MG INJ be stored prior to use?
A: The product should be stored in a refrigerator at a temperature between 2C and 8C. Avoid freezing the vial, as this may damage the medication and reduce its effectiveness.Q: What steps are involved in the reconstitution process?
A: A healthcare provider will mix the lyophilized powder in the vial with the supplied sterile water for injection according to standardized protocols. The reconstituted solution will be gently swirled (not shaken) and checked for particulate matter or discoloration before administration.Q: How does BRYXTA 400MG INJ benefit patients with HER2-positive cancers?
A: It targets HER2 receptors on cancer cells, helping to inhibit their growth and survival. This targeted approach provides significant clinical benefits, including improved progression-free survival and response rates for qualifying patients.Q: Is BRYXTA 400MG INJ suitable for home administration?
A: No, due to the need for precise reconstitution and intravenous infusion, BRYXTA 400MG INJ must be administered by healthcare professionals in a controlled medical setting.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email



 Contact Us
Contact Us Call Me Free
Call Me Free
